
The Swine Health Information Center funded a study to evaluate tongue fluids (TF) samples from stillborn pigs as an indicator of PRRS detection in liveborn littermates. Led by Drs. Isadora Machado and Daniel Linhares of Iowa State University, the study investigated TF as a risk-based sample type in commercial breeding herds to predict PRRSV detection in the litter. Litters with stillborn piglets and small litter size had a higher probability of being PRRSV positive. Moreover, TF from stillborn piglets were shown to be a reliable indicator of PRRSV status for their liveborn littermates, indicating that TF can be an effective risk-based approach for PRRSV detection.
Find the study industry summary for project #23-076 on this page.
A risk-based approach to animal selection for sampling enhances PRRSV detection by increasing the probability of selecting an animal harboring the pathogen while requiring a smaller sample size. Aggregated TF from dead animals has emerged as a promising risk-based approach. It has a similar PRRSV RNA positivity rate as serum, processing fluids, and family oral fluids. Its composition is complex, especially in stillborns, consisting of blood, saliva, respiratory mucus, amniotic fluid, meconium, and environmental contaminants, which may reflect the litter’s PRRSV status upon laboratory testing.
Objectives of the study described herein had a goal to further knowledge on the use of TF from stillborns in commercial sow herds for PRRSV detection. Objectives included evaluating 1) the incidence of stillborn piglets on the probability of PRRSV RNA detection within the litter, 2) the stillborn TF PCR-results as an indicator of PRRSV in liveborn piglets, and 3) the litter size as a risk factor for PRRSV
To investigate these objectives, samples were collected from two PRRSV-positive breeding herds (Herd A: 2,500-sow farm with a three-week batch farrowing system; Herd B: 4,500-sow farm with a weekly batch farrowing system). A total of 130 litters were sampled within 12 hours after farrowing: 66 litters without stillborn piglets and 64 litters with stillborn piglets. This totaled 1,723 liveborn and 105 stillborn piglets sampled. Regarding the litters’ parity, in Herd A, 14 were parity 1 sows, seven were parity 2, four were parity 3, and one was parity 4. For Herd B, all 104 litters came from parity 1 sows.
TF and intracardiac blood samples were individually collected from stillborn piglets. Tail blood swabs were individually collected from all liveborn littermates. Samples were individually tested for PRRSV RNA detection by RT-qPCR at the Iowa State University Veterinary Diagnostic Laboratory. Litters with ≤ 11 liveborn piglets were defined as small litters, whereas litters with ≥ 12 were defined as large.
Across all sample types, the percentage of PRRSV-positive litters was 22.3% (29/130 litters), of which 15.4% (20/130 litters) were tail blood swab PCR positive, 10% (13/130 litters) were intracardiac blood PCR positive, and 16.9% (22/130 litters) were TF PCR positive. At an individual animal-level across both herds, 3.4% (59/1,723 liveborn piglets) were tail blood swab PCR positive, 16.2% (17/105 stillborn piglets) were intracardiac blood PCR positive, and 26.7% (28/105 stillborn piglets) were TF PCR positive. The mean positivity of liveborn piglets within the litter was 5% but varied between 0 – 85.7%, while the total born (stillborn and liveborn piglets combined) was 4.6% (0 – 76.4%).
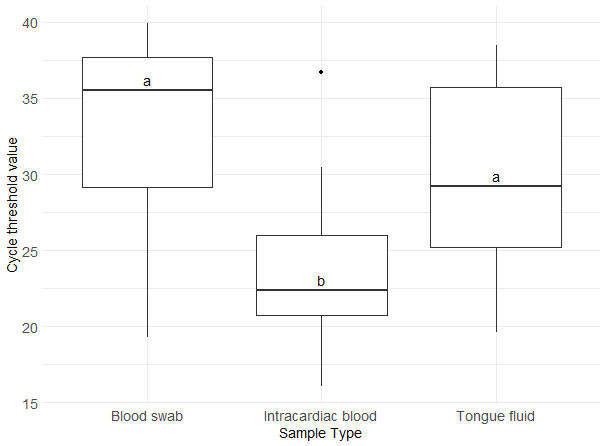
Regarding sample type Ct values, tail blood swabs had a median of 35.5 (interquartile range, IQR: 29.2-37.7), intracardiac blood a median of 22.4 (IQR: 20.7-26.0), and TF a median of 29.2 (IQR: 25.2-35.7) (Figure 1). Intracardiac blood exhibited the lowest median Ct value, which was significantly different from the median Ct values of the other sample types (P < 0.05).
In the model analysis, litter size and the presence of stillborn piglets were used as predictor variables, while litter PRRSV status was the response variable. Small litters had 12.2 times higher odds (P < 0.001) of having a PRRSV-positive piglet compared to large litters. Holding stillborn presence constant, there was a 51.9% probability of detecting PRRSV-positive piglets in small litters compared to 8.1% in large litters. Similarly, litters with at least one stillborn piglet had 12.5 times higher odds (P < 0.001) of having a PRRSV-positive piglet compared to litters without stillborn piglets. Holding litter size constant, there was a 52.2% probability of detecting a PRRSV-positive piglet when at least one stillborn was present, compared to 8.0% when no stillborns were present. No significant interaction between litter size and stillborn presence was found (P = 0.076).
When considering at least one TF PRRSV-positive result and litter size as predictors to assess the odds of having a PRRSV-positive liveborn littermate, litters with at least one TF-positive result had 17.6 times higher odds (P < 0.001) of having a PRRSV-positive liveborn piglet compared to litters without stillborn piglets. Holding litter size constant, there was a 63.6% probability of detecting PRRSV-positive liveborn piglets when at least one TF result is PRRSV-positive compared to 9.0% when stillborn were absent or 4.0% when TF result was PRRSV-negative. Similarly, small litters had 7.0 times higher odds (P = 0.002) of having a PRRSV-positive liveborn piglet compared to large litters. Holding the TF results constant, there was a 20.8% probability of detecting a PRRSV-positive liveborn littermate in small litters compared to 3.6% in large litters.
In conclusion, litters with stillborn piglets and small litter size have a higher probability of detecting PRRSV in the farrowing room. Moreover, TF from stillborn piglets are a reliable indicator of the PRRSV status of their liveborn littermates, indicating that TF can be an effective risk-based approach for PRRSV detection.
Veterinarians and producers are encouraged to collect TF, targeting stillborn piglets and small litters to increase the likelihood of detecting PRRSV. This risk-based sampling strategy within the first few hours post-farrowing enhances the effectiveness of PRRSV monitoring programs in breeding herds and supports timely interventions, such as McREBEL and cross-fostering. Lastly, while TF were individually collected in this study to test the hypothesis, collecting them as an aggregated sample, similar to PF, is recommended. This approach increases the likelihood of detecting the virus, allowing for a larger number of animals to be screened.
Copyright 2025 | Swinehealth.org | Website by Heartland Marketing Group